Square Planar Mo Diagram. Of these 3 are bonding and hence 3 are antibonding leaving 1 non bonding orbital. In this video i have explained the detailed molecular orbital diagram for square planar complexes.
Why is p d c l x 4 x 2 square planar if c l is not a strong field ligand. Nh 3 4 aos on n and 3 for 3h gives 7 mos to consider. To see why we should consider nickel which is in the same group whose complexes are tetrahedral sometimes and square planar other times.
Walsh diagrams summarise changes in mo diagram wrt structure note a combination of first and second order effects.
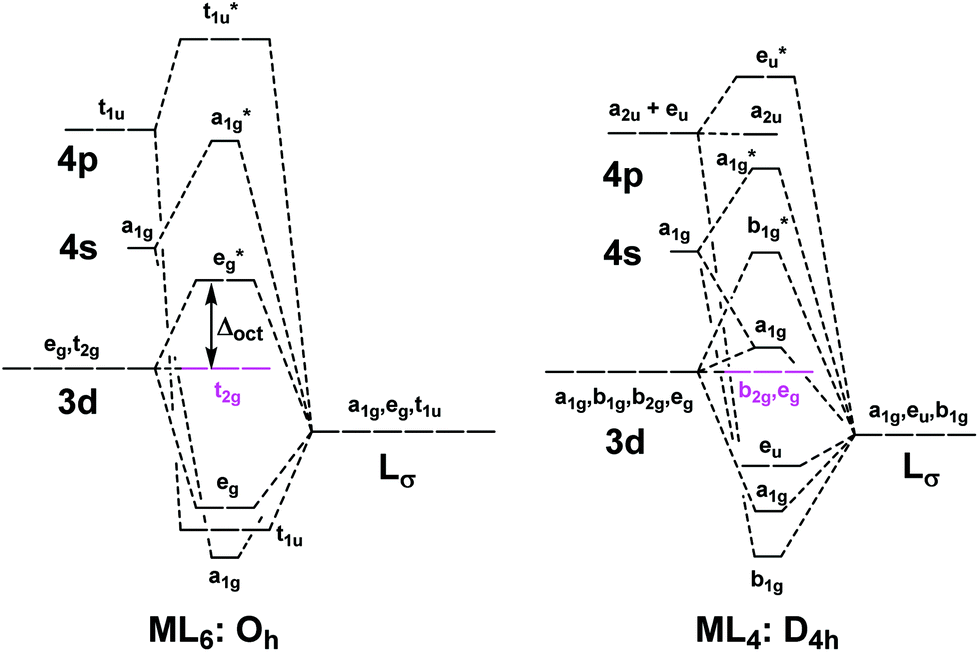
Bond square planar tetrahedral. The square planar geometry is prevalent for transition metal complexes with d 8 configuration. Tetragonal elongation removal of z ligands eg t2g b2g dxydxzdyz eg dz2 dx2 y2 dxzdyz dxy dz2 dx2 y2 a1g b1g b2g eg dxzdyz dxy dz2 dx2 y2. Why exactly is the planar ch4 molecule unstable relative to the tetrahedral arrangement.